 Developing Breakthrough Therapies for
Rare Inflammatory Diseases
www.CorbusPharma.com
OTCQB: CRBP
Exhibit 99.1 |
 Developing Breakthrough Therapies for
Rare Inflammatory Diseases
www.CorbusPharma.com
OTCQB: CRBP
Exhibit 99.1 |
 2
Forward-Looking
Statement
2
This presentation contains certain forward-looking statements, including those relating to the
Company’s product development, clinical and regulatory timelines, market opportunity,
competitive position, possible or assumed future results of operations, business strategies,
potential growth opportunities and other statement that are predictive in nature.
Additional written and oral forward-looking statements may be made by the Company from time
to time in filings with the Securities and Exchange Commission (SEC) or otherwise. The
Private Securities Litigation Reform Act of 1995 provides a safe-harbor for forward-looking
statements. These statements may be identified by the use of forward-looking expressions, including, but not
limited to, “expect,” “anticipate,” “intend,” “plan,”
“believe,” “estimate,” “potential,” “predict,” “project,” “should,” “would,”
“will” and similar expressions and the negatives of those terms. These statements
involve known and unknown risks, uncertainties, and other factors which may cause actual
results, performance or achievements to be materially different from any future results,
performance or achievements expressed or implied by the forward-looking statements.
Prospective investors are cautioned not to place undue reliance on such forward- looking
statements, which speak only as of the date of this presentation. The Company undertakes no
obligation to publicly update any forward-looking statement, whether as a result of new
information, future events or otherwise. |
 3
Overview
•
Corbus Pharma is focusing on rare, life-threatening, chronic inflammatory
diseases •
Lead drug Resunab™: a first-in-class oral
anti-inflammatory/fibrosis small molecule •
Acts to trigger inflammatory resolution: the “off”
switch for chronic inflammation
•
Proven safe in Phase 1 + promising pre-clinical potency in multiple animal
models •
Phase 2 clinical trials to commence 2015:
•
Successful $10.3m private financing round (May 2014)
•
Obtained $1.3m in NIH grants
•
IP protection until 2033 and potentially longer
•
Commenced trading on OTC.QB in October 2014
3
•
Cystic Fibrosis (CF)
•
Diffuse Systemic Sclerosis (SSC) also known as “Scleroderma”
|
 Our
Target Indications: Current & Future Indication
Patient numbers
(USA)
Estimated
Market size
Current therapies for
inflammation
Drawbacks to current
therapies
Current lead indications:
Cystic Fibrosis
30,000
>$3B
Steroids, ibuprofen
Considerable side effects
Diffuse Systemic
Sclerosis
(Scleroderma)
50,000
>$2B
Steroids,
methotrexate
Side effects, poor efficacy
Potential future indications:
Dermatomyositis
13,000
>$1B
Steroids, mAbs
Side effects, poor efficacy
Marfan Syndrome
5,000
>$1B
N/A
Lupus (SLE)
500,000-1.5MM
>$3B
Steroids, mAbs
Side effects, poor efficacy
Idiopathic Pulmonary
Fibrosis (IPF)
70,000
>$1B
Pirfenidone
Limited efficacy
InterMune bought by Roche
for $8.5B (2014)
4 |
 5
Inflammation
“ON”
Pro-inflammatory
Mediators
(IL6, IL8, PGE2, LTB4)
Immune cell
TGF-ß
CB2 receptor
Inflammation
“OFF”
Fibrosis
“ON”
Fibrosis
“OFF”
•
CB2 receptor is present on immune cells and activated by endogenous lipid
mediators •
Activation of CB2 turns inflammation off (“inflammatory
resolution”) •
Resunab expected to be first CB2-binding anti-inflammatory drug to
reach market •
Upstream of other approaches: potential for better safety and potency
CB2 Receptor: Turns inflammation “off”
Immune cell
5
COX
LOX
LXA
Anti-
inflammatory
Mediators
(Lipoxins,
Resolvins, etc)
4 |
 6
|
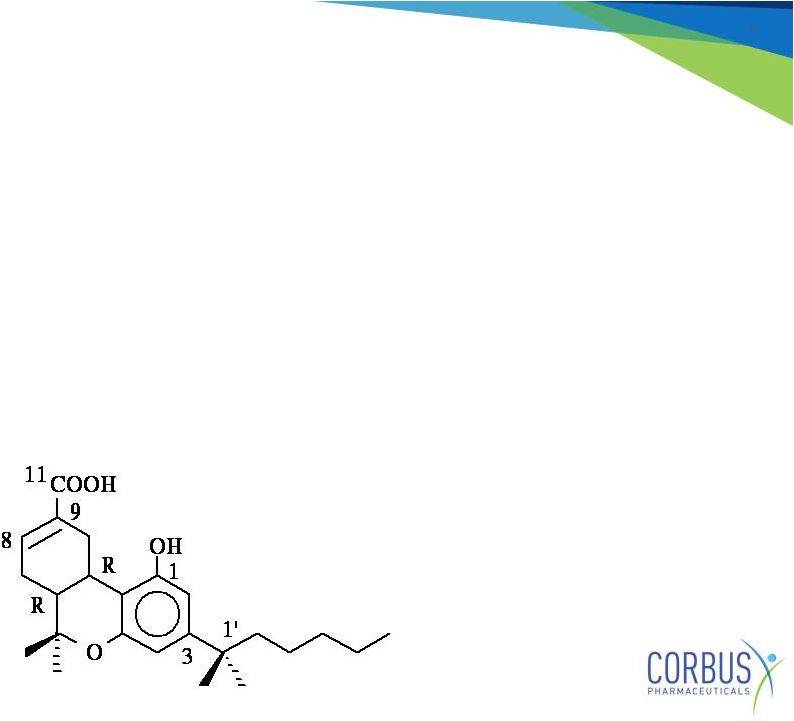 7
Resunab
7
•
Resunab: synthetic oral CB2 agonist small-molecule
•
Designed to trigger the resolution of chronic inflammation
•
Full manufacturing, drug supply, non-clinical safety & pharmacology
package for Phase 2 programs
•
Excellent clinical safety profile to date: two prior Phase 1 clinical trials
(n=121) •
Preparing to launch two Phase 2 clinical studies in 2015
•
Lacks
CNS
side
effects
of
other
CB2-binding
class
members
•
Lacks GI side effects of NSAID’s (e.g. Aspirin™, ibuprofen,
Celebrex™) •
Lacks metabolic side effects of corticosteroids |
 Resunab: Only
CB2-Agonist
Targeting
Inflammation
Company
Indication
Brain penetration
Status
Affects CNS
Corbus Pharma
Inflammation
Minimal
Entering
Phase 2
No
AbbVie
Pain
Full
Phase 1
Yes
Glenmark
Pain
Full
Phase 1
Yes
Eli Lilly
Knee pain
Full
Phase 2
Yes
AstraZeneca
Post operative pain
Full
Phase 2
Yes
Resunab is the only CB2 drug that can be used to treat
inflammation because it does not target the brain
8 |
 9
Cystic Fibrosis
Targeting inflammation at the core of the disease
9 |
 10
Antibiotics
Kalydeco
(for 6%) of
patients
Very limited
treatment
No treatment
Overview: Cystic Fibrosis
10
•
Inflammatory orphan disease (30,000 patients in USA, 75,000 WW)
•
Average life expectancy of CF patients is approximately 40 years
•
Inflammation at core of disease‘s morbidity and mortality (pulmonary
fibrosis) •
Very high doses of steroids/ibuprofen effective but rarely used due to
toxicity •
Need for safe, chronic anti-inflammatory drug is unmet and universally
recognized •
Pharmaco-economics support premium pricing (e.g. Kalydeco by Vertex priced
at $320,000/yr) CFTR gene
defect
Infection
Fibrosis
Underlying
Inflammation |
 11
Antibiotics
Kalydeco
(for 6%)
Resunab
Resunab
Resunab
targets key CF inflammatory players
11
TGF-
Lipoxin-A4
•
•
•
Absent in CF lungs
•
Genetically linked to disease
Associated with worsening symptoms
Replacement therapy effective in
animal models
CFTR defect
Infection
Fibrosis
Underlying
Inflammation |
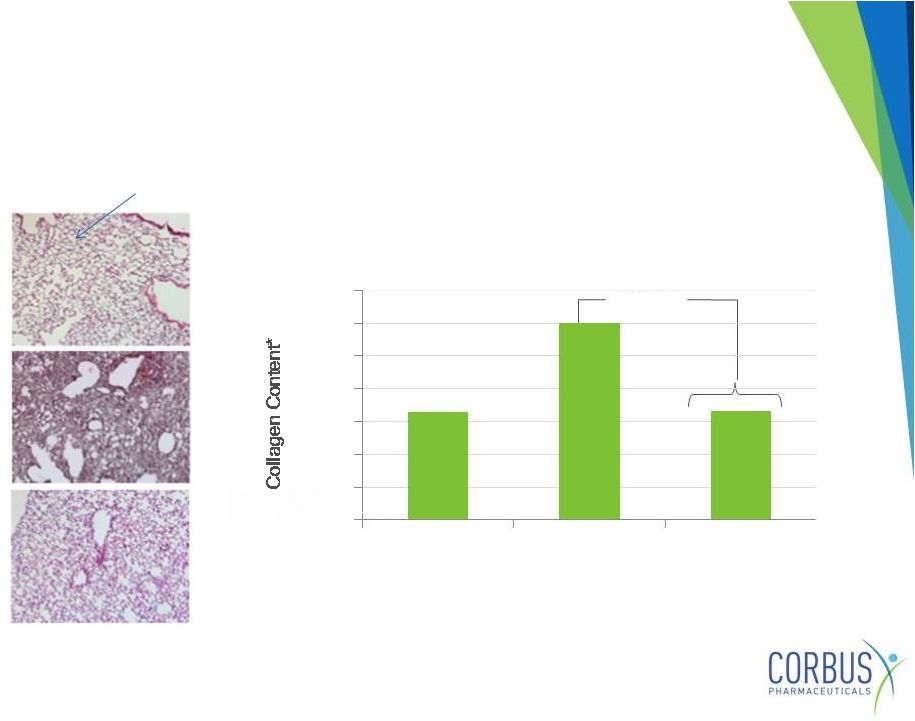 Fibrosis-inducing agent (Bleomycin) administered to lungs
day 1 followed by daily oral Resunab for 21 days
P<.004
Bleomycin+
Resunab
therapy
Bleomycin
Control
Gonzales et.al., Annals of Rheumatic Diseases, 2012. 71:1545-51
* Measured by hydroxyproline
Alveoli -air sacs
Resunab
Reduces Pulmonary
Fibrosis In Animal Models
Control
Bleomycin
Bleomycin
+
Resunab
12
0
50
100
150
200
250
300
350 |
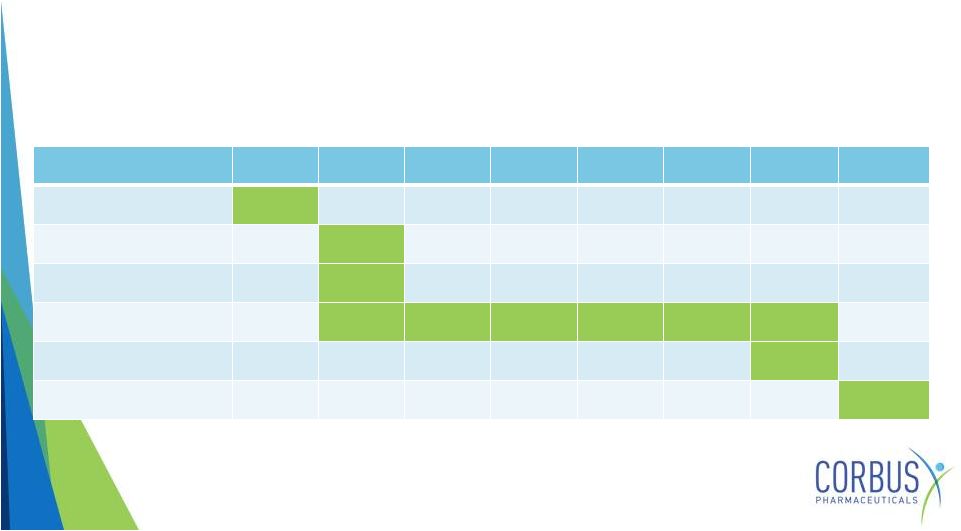 Resunab
Planned Cystic Fibrosis Phase 2 Trial
13
Q1 2015
Q2 2015
Q3 2015
Q4 2015
Q1 2016
Q2 2016
Q3 2016
Q4 2016
Protocol filed with FDA
X
Study launches
X
First patient dosed
X
Study duration
X
X
X
X
X
X
Last patient dosed
X
Study data released
X
•
Double blind placebo control study in the USA under IND from FDA
•
Primary
end
points:
Safety/tolerability
+
PK
•
Secondary
end
points:
Inflammatory
biomarkers+
MOA
+
change
in
clinical
outcome
measures
•
Patient
number:
70
adults
with
CF
in
15-20
sites
•
Treatment
duration:
3
months
+
1
month
follow-up
•
Dose
response:
1mg/day,
5mg/day,
20mg/day
and
20mg/2Xday |
 14
Diffuse Systemic
Sclerosis
(“Scleroderma”)
Relief for a disease with no
effective long-term therapy |
 Overview: Diffuse Systemic Sclerosis (Scleroderma)
15
•
Chronic inflammatory disease causing fibrosis of skin, joints and internal
organs •
Orphan disease (50,000 patients in USA)
•
80% of patients are women in their 30’s and 40’s
•
Common cause of death: lung fibrosis (50% mortality in 10 years)
•
Early stage of disease responds to steroids/methotrexate but with serious side
effects •
No effective and safe long-term therapy available
•
Pipelines often target Idiopathic Pulmonary Fibrosis (IPF) in conjunction to
SSC |
 16
Resunab
Inhibits Key Factors in SSC
•
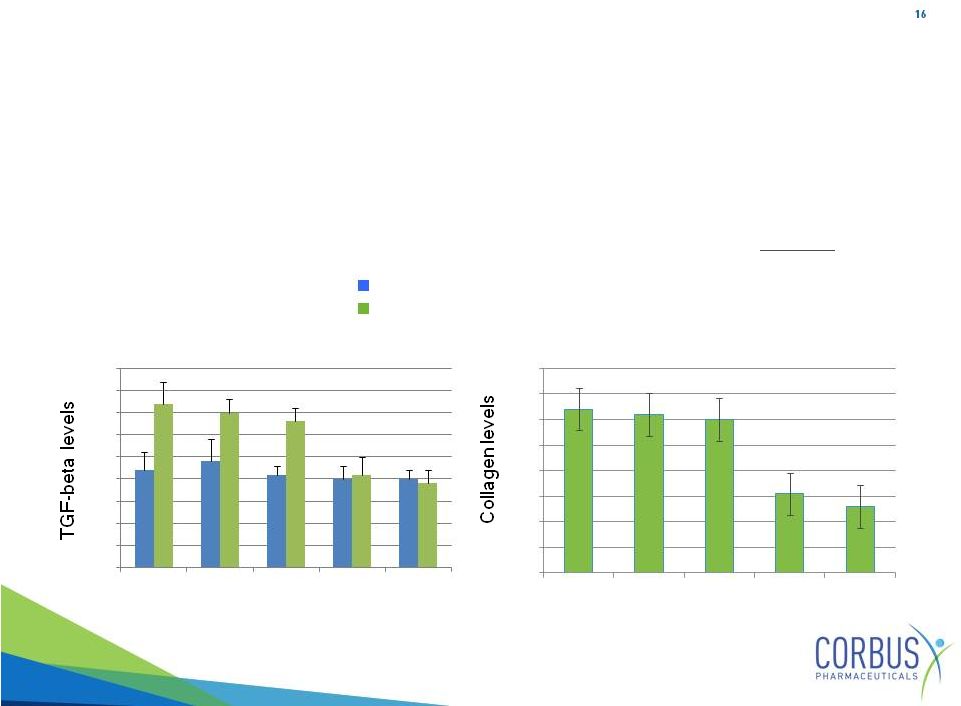
TGF-beta plays key role in SSC progression (same in CF and IPF)
•
Elevated TGF-beta levels associated with disease progression
•
Strong Resunab efficacy data in animal models
•
Resunab
reduces
TGF-beta
and
collagen
in
skin
fibroblasts
from
SSC
patients
TGF-beta
Collagen
Concentration of Resunab
(µM)
Concentration of Resunab
(µM) Healthy skin
fibroblasts Scleroderma patients’
skin
fibroblasts
0
5
10
15
20
25
30
35
40
45
0
0.1
1
5
10
0
50
100
150
200
250
300
350
400
0
0.1
1
5
10 |
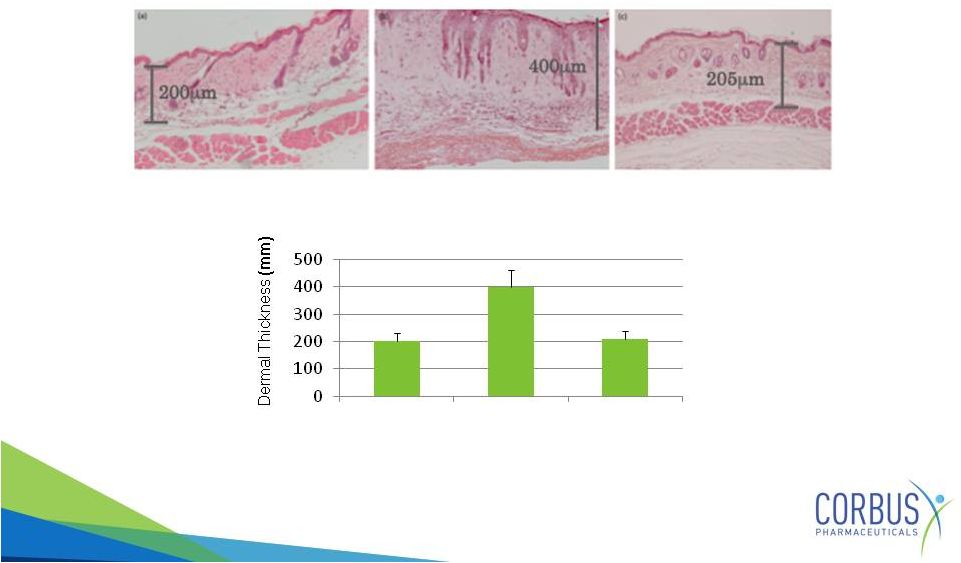 Resunab
Inhibits Skin Thickening
In Mouse SSC Model
Gonzales et.al., Annals of Rheumatic Diseases, April 4,
2012
17
Control
Bleomycin
Bleomycin
+
Resunab
Thick skin induced by
Bleomycin
Near normal skin after oral
Resunab
taken once daily
for four weeks
Healthy skin |
 Resunab:
Planned
SSC
Phase
2
Clinical
Trial
18
Q1 2015
Q2 2015
Q3 2015
Q4 2015
Q1 2016
Q2 2016
Q3 2016
Q4 2016
Protocol filed with
FDA
X
Study launches
X
First patient dosed
X
Study duration
X
X
X
X
X
X
Last patient dosed
X
Study data released
X
•
Double blind placebo control study in USA under IND from FDA
•
Primary
end
points:
Safety/tolerability
+
PK+
Change
in
clinical
outcomes
•
Secondary
end
points:
Inflammatory
biomarkers+
quality
of
life
(QOL)
•
Patient
number:
35
adults
with
SSC
with
8-10
sites
•
Treatment
duration:
3
months
+
1
month
follow-up
•
Dose
response:
5mg/day,
20mg/day
and
20mg/2Xday |
 Management Team
Yuval Cohen, Ph.D.- Chief Executive Officer
Mark Tepper, Ph.D.- President & Chief Scientific Officer
Sean Moran, C.P.A. M.B.A.- Chief Financial
Officer Barbara White,
M.D.- Chief Medical Officer
•
Co-founder and former President of Celsus Therapeutics (CLTX)
•
Expertise in developing anti-inflammatory drugs including for CF
•
Former VP USA Research & Operations, EMD Serono; Sr. Investigator,
Bristol-Myers Squibb •
Key member of project teams which developed the following marketed drugs:
Taxol® (Ovarian Cancer, 2000 peak sales of $1.6B),
Orencia® (RA, 2013 sales of $1.4B), Rebif®
(MS, 2013 sales of $2.59B), Gonal-F®
(Fertility, 2013 sales of $815MM)
•
Former CFO: InVivo (NVIV), Celsion (CLSN), Transport Pharma, Echo Therapeutics
(ECTE) & Anika Therapeutics (ANIK)
•
Former VP Clinical & Medical Director at Amgen, UCB and Medimmune. Expert
rheumatologist in scleroderma with decades of experience in clinical
trial development. 19 |
 Board
of Directors Former CEO of PhRMA (1996-2005)
Over
two
decades
of
public
service
in
Washington,
D.C.
including
Special
Envoy
to
China
(2007-2009)
Former board member Inspire Pharma (sold to Merck for $430m in 2011)
Chairman of the Board of the Metropolitan Washington, D.C. Chapter of the
Cystic Fibrosis Foundation Managing Partner of Orchestra Medical
Ventures Over 17 years of venture capital and investment banking
experience Former Managing Director of Spencer Trask Ventures, Inc.
securing over $420 million in equity capital 25 years of development,
regulatory and senior management experience in the biopharm industry
Former
CMO
of
Insmed,
a
specialty
CF
company
and
current
advisor
to
the
CEO
Former Vice President and Head of US Clinical Research and Development at
Novartis (2003-2006) CFO Celldex Therapeutics (CLDX) since 2000
Raised over $415MM financing
20 years experience in industry: Repligen (CFO) and Endogen (CFO)
20
Yuval Cohen, Ph.D.- Chief Executive Officer
Amb. Alan Holmer – Chairman of the Board
David Hochmann
Renu Gupta, MD
Avery W. (Chip ) Caitlin |
 World
Class Scientific Advisors Professor of Biochemistry and Pharmacology;
inventor of Resunab Professor of Pulmonary and Critical Care
Medicine Professor Medicine, National PI on largest ever
anti-inflammatory CF study Chairman of International Clinical
Scleroderma Consortium Director of UCLA Scleroderma Program
Ex-Chair of Rheumatology
21
Sumner Burstein, Ph.D. - UMass Medical School
Michael Knowles, M.D., Ph.D. - UNC Chapel Hill
James Chmiel, M..D. – Case Western Reserve Medical School
Robert Simms, M.D. - Boston University School of Medicine
Daniel Furst, M.D. – UCLA School of Medicine Robert Zurier,
M..D. - UMass Medical School |
 22
Financial Profile
22
Stock Ticker:
CRBP:OTCQB
$77,400,000
Market
capitalization
as
of
November 5,
2014
$10,300,000
Raise from successful private placement (Q2 2014) from institutional and retail base
25,800,000
Common shares outstanding
41,500,000
Fully diluted shares outstanding (including warrants and stock options)
$11,400,000
Available from exercise of callable warrants
NASDAQ
Up-listing to NASDAQ planned by Q-1 2015
Q1 2015
File IND
with FDA
Q1+Q2
2015
Launch
clinical
trials
Q1 2015
Plan to
up-list to
NASDAQ
H2 2016
Data from
clinical
trials
Q4 2014
Q4 2016 |
 23
Corbus Poised for Significant Upside
Recent Deals
Date
Company
Partner
Type
Drug
Indication
Stage
Up-Front
Deal Total
11/14
Galecto
BMS
Option to
acquire
TD139
Idiopathic pulmonary fibrosis
Phase 1
NA
$444M*
8/14
InterMune
Roche
Acquisition
Esbriet
Idiopathic pulmonary fibrosis
Approved
NA
$8.3B*
9/2013
Galapagos
AbbVie
License
GLPG1837
Mutations in CF patients,
including F508del and G551D
Pre-clinical
$45M*
$405M*
7/2011
Amira
BMS
Acquisition
AM152
Idiopathic pulmonary fibrosis
and systemic sclerosis
Phase 1
$325M*
$475M*
* Figures from company press releases
$405
$444
$450
$8,300
$100
$1,000
$10,000
Galapagos
(License -
AbbVie)
Galecto
(M&A -
BMS)
Amira
(M&A -
BMS)
Intermune
(M&A -
Roche)
Recent Deals in CF and Fibrosis
Pre-Clinical
Phase 1
Phase 3 |
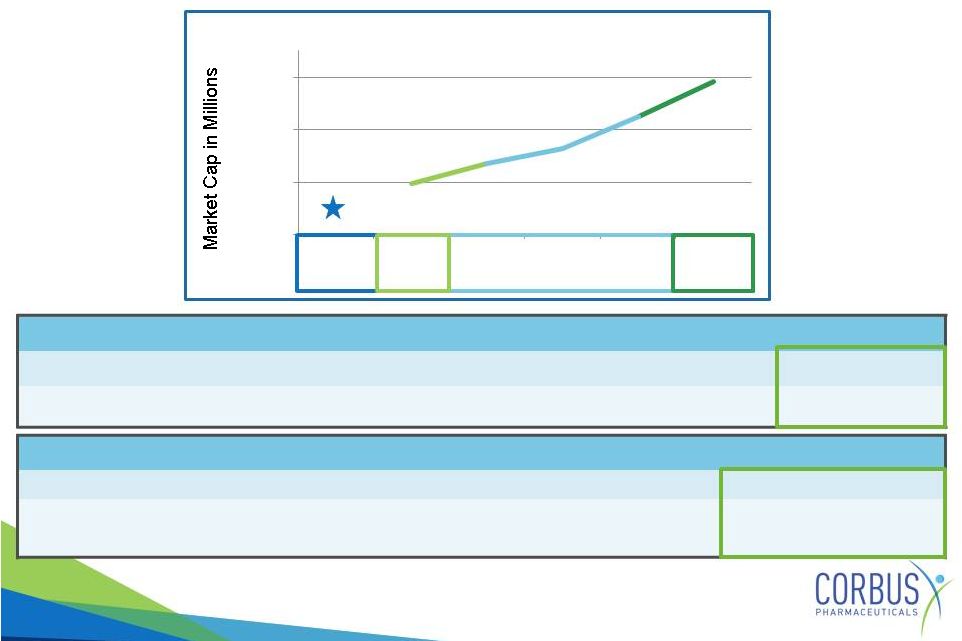 Potential Value Indicators
Recent IPO
Date
Company
Lead Compound
Indication
Stage
Market Cap
9/2014
ProQR
QR-010
Cystic
Fibrosis
-
RNA
repair
Pre-clinical
$284.11M
Company
Drug
Indication
Cost per Year
2018 Sales Est.
Vertex
Kalydeco
Cystic Fibrosis -
mutations of CFTR gene: G551D, G1244E, G1349D,
G178R, G551S, S1251N, S1255P, S549N, or S549R
$294,000
$1.2B**
* Figures from company press releases
** Leerink analyst report
Market Cap of CF and Fibrosis Focused Companies
24
$286
$685
$1,347
$5,335
$26,000
$77
30
300
3000
30000
Corbus
ProQR
Insmed
PTC
Intercept
Vertex
Market
Phase 3
PC
Phase 2a
Approved Products |
 Conclusions
•
Lead Product Resunab
is a novel, safe and promisingly potent clinical
stage anti-inflammatory/anti-fibrotic drug which acts to resolve
inflammation
•
Targets multiple rare inflammatory indications
•
Proven safe in two Phase 1 trials
•
Promising potency in multiple pre-clinical models
•
Launch two Phase 2 trials in 2015 (Cystic Fibrosis and Scleroderma)
•
Completion of studies in 2016
•
Strong patent portfolio until 2033
25 |
 26
100 River Ridge Drive
Norwood, MA 02062
www.CorbusPharma.com |